
are usually called Elements, of which all other Bodies are compounded: Elements are such Substances as cannot be resolved, or reduced, into two or more Substances of different Kinds. Īmong Substances, some are called Simple, some are Compound. In his Logick, published in 1724, the English minister and logician Isaac Watts gave an early definition of chemical element, and contrasted element with chemical compound in clear, modern terms. Isaac Watts Portrait of Isaac Watts by John Shury, c. wayes, that an almost incredible number of. Corpuscles may be mingled in such various Proportions, and. If we assigne to the Corpuscles, whereof each Element consists, a peculiar size and shape. Corpuscles of elements and compoundsīoyle used the concept of "corpuscles"-or "atomes", as he also called them-to explain how a limited number of elements could combine into a vast number of compounds: And yet out of all these exotick Compounds, we may recover the very same running Mercury. with Sulphur it will compose a blood-red and volatile Cinaber. While the distinction between compound and mixture is not so clear, the distinction between element and compound is a central theme. "Perfectly mixt bodies" included for example gold, lead, mercury, and wine. 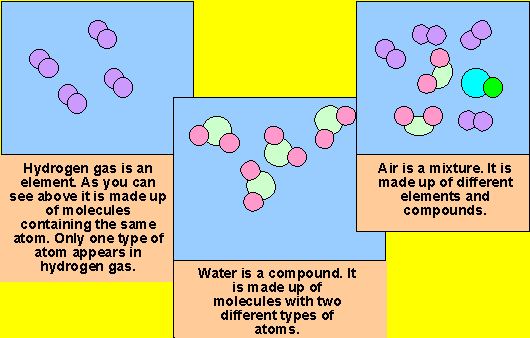
In this book, Boyle variously used the terms "compound", "compounded body", "perfectly mixt body", and "concrete". The term "compound"-with a meaning similar to the modern-has been used at least since 1661 when Robert Boyle's The Sceptical Chymist was published. History of the concept Robert Boyle Robert Boyle Title page of The Sceptical Chymist Globally, more than 350,000 chemical compounds (including mixtures of chemicals) have been registered for production and use. Many chemical compounds have a unique CAS number identifier assigned by the Chemical Abstracts Service. Non-stoichiometric compounds form a disputed marginal case.Ī chemical formula specifies the number of atoms of each element in a compound molecule, using the standard chemical symbols with numerical subscripts. Molecular compounds are held together by covalent bonds ionic compounds are held together by ionic bonds intermetallic compounds are held together by metallic bonds coordination complexes are held together by coordinate covalent bonds. There are four major types of compounds, distinguished by how the constituent atoms are bonded together. In this process, bonds between atoms may be broken and/or new bonds formed. A compound can be transformed into a different substance by a chemical reaction, which may involve interactions with other substances. /periodic-table-of-the-elements-2017--illustration-769723031-5aa02f9b04d1cf00386ccf7c.jpg)
A molecule consisting of atoms of only one element is therefore not a compound. The ball-and-stick model of the molecule shows the spatial association of two parts hydrogen (white) and one part oxygen (red)Ī chemical compound is a chemical substance composed of many identical molecules (or molecular entities) containing atoms from more than one chemical element held together by chemical bonds. Pure water (H 2O) is an example of a compound. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
